How molecular signatures of food intake in human stool can transform the way we collect and analyze dietary data
Current methods for dietary assessment in nutrition research depend on asking people to self-report what they eat, which face challenges including participant compliance, accurate recall, and the inability to capture country-specific food diversity. To address these issues, Lawrence David, PhD, associate professor from Duke University (USA), developed FoodSeq, a strategy termed “DNA metabarcoding” that leverages DNA sequencing technology to measure diet from degraded food DNA in human stool samples. The number and types of plants consumed detected by metabarcoding correlates with dietary diversity and quality estimates from established self-report assessments, age, and household income1.
David also presented case studies showing how this genomic approach can be used for studies of global and population health, including the identification of ultra-processed foods using plant species richness and analysis of food DNA in wastewater, the later acting as a proxy of dietary relationships with economic status, place of living (urban/rural), and culture. Some limitations of metabarcoding are that highly absorbable food constituents do not show in fecal food DNA and the inability of DNA to distinguish food type for foods derived from the same source species (e.g., chicken and egg), which can be solved by integrating metaproteomics.
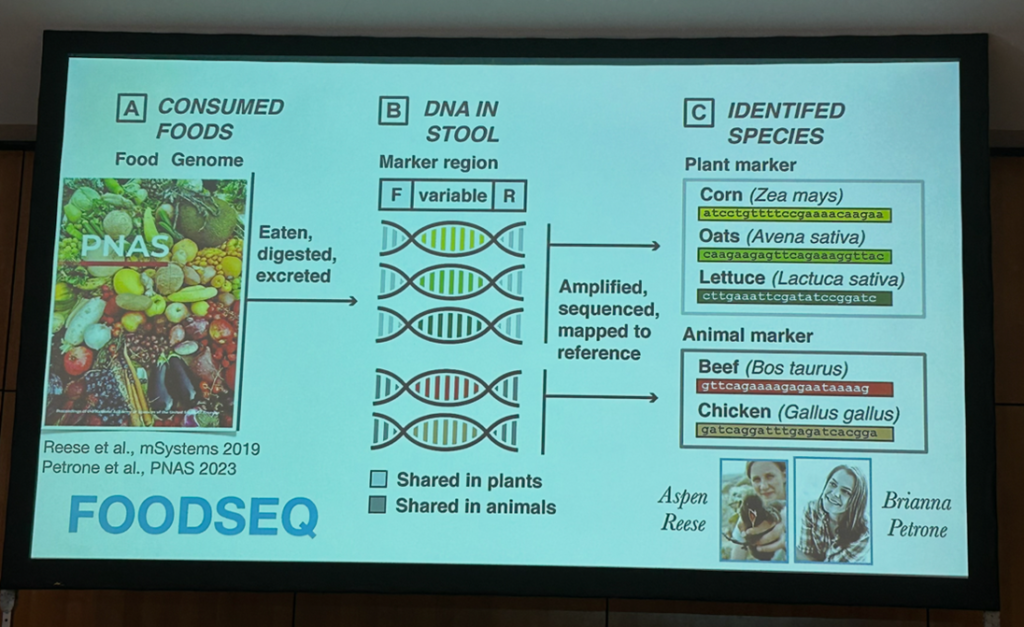
Molecular signatures of food intake in human stool capture dietary plant diversity. Source: David’s presentation at the GMFH World Summit 2026.
The effects of ultra-processed foods and fiber in the gut may differ in the presence of disease and an altered gut microbiome
Ciarán G. Forde, PhD, from Wageningen University (The Netherlands) presented findings on the RESTRUCTURE randomized controlled trial that consuming over 90% of daily calories from ultra-processed foods (UPFs) for 14 days did not have an effect on gut health markers, gut microbiota composition, diversity and functionality, or inflammatory markers in healthy individuals. These findings should be taken cautiously as the 14-day window may be too brief to detect meaningful changes in microbiome endpoints. Moreover, not all food additives impact the same way the gut microbiome2 and the ADDapt trial showed that emulsifier dietary restriction for 8 weeks can reduce symptoms and fecal calprotectin in patients with active Crohn’s disease3, so the impact of UPFs on the gut may differ between healthy and clinical populations and from person to person. Also, the speed of digestion of individual food constituents is a confounder and as gut microbiome modulation occurs via mucus alteration and gut permeability, these outcomes should be considered when interpreting findings.
Heather Armstrong, MSc, PhD, from University of Manitoba (Canada), debunked the myth that all dietary fibers are beneficial. Armstrong presented recent findings on the importance of personalizing dietary fiber intake, especially during disease. Some types of fiber such as b-fructans may induce inflammation and trigger flares in a subset of patients with IBD4 and worsen demyelination and promote gut-brain axis immune activation in mice5 if the microbes needed to ferment the fiber are not present.
Armstrong explained that consuming some pectins with b-fructans (even when b-fructans are not fermented) may inhibit inflammation in IBD models through binding toll-like receptors. Beyond fiber type and quantity, fiber matrix also matters, with emerging findings in mice and isolated case reports showing high levels of inulin promoting colon and liver cancer6 and inflammation-promoting effects of arabinoxylans at higher doses7. Armstrong is working on a clinical trial to identify, based on fecal microbiota signature and other factors, which carbohydrates are beneficial, neutral or harmful for people with IBD and other inflammatory illnesses and people with normal digestion. In the meanwhile, Armstrong acknowledged that it is key to go slow and low with fiber to allow the colon microbiome to adapt to the fiber change.

Upcoming clinical trial to test what specific types of carbohydrates are best for healthy individuals and those with IBD based on their gut microbiota profile. Source: Heather Armstrong’s presentation at the GMFH World Summit 2026.
Jens Walter, PhD, professor of ecology, food, and the microbiome at University College Cork (Ireland), presented findings on the benefits of a non-industrialized-type diet on the gut microbiome and on the cardiometabolic risk in 30 healthy Canadian adults. The high-fiber, mostly plant-based diet, termed NiMe diet, was associated with a decrease in body weight, fasting cholesterol levels and C-reactive protein, which were linked to changes in the gut microbiome affected by industrialization8. Walter also showed that the benefits of high-fiber, whole food diets on immunometabolic markers of chronic diseases in healthy adults were not recapitulated in >80% interventions with isolated and synthetic dietary fiber9. Effects of fiber on health outcomes are fiber structure-specific and are linked to gut microbiome shifts. In adults with excess body weight, a 6-week randomized controlled trial showed that baseline gut microbiome features may predict clinical responses to dietary fibers. Machine learning models predicted individualized, fiber-specific effects on diastolic blood pressure (acacia gum) and C-reactive protein (resistant starch 4) from microbial pentose phosphate pathway and fecal bile acids. Interestingly, effects on markers of inflammation, barrier function, and satiety also occurred via microbiome-independent mechanisms10.
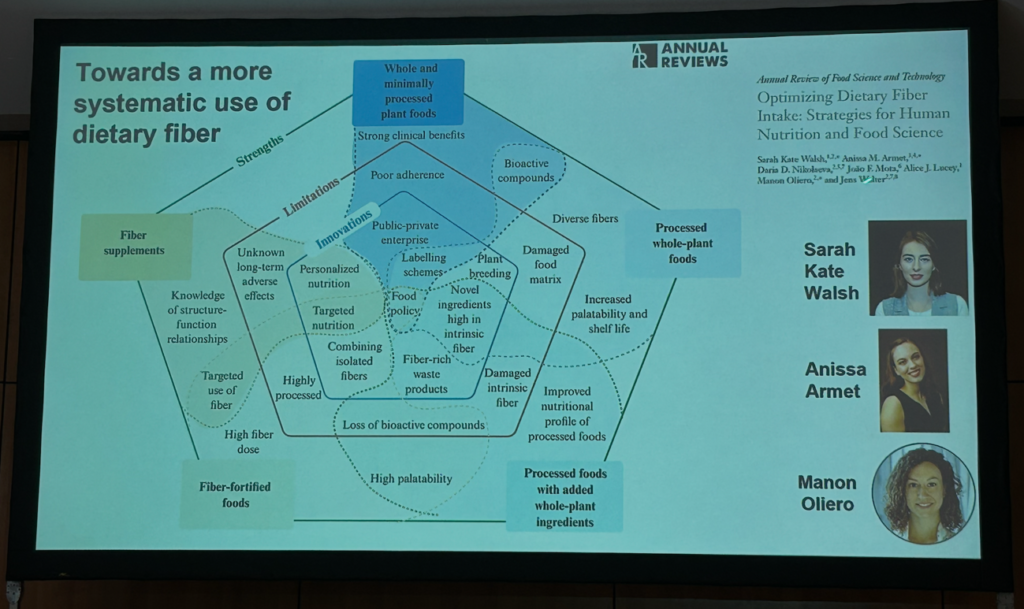
Strengths and limitations of fiber-based nutrition interventions and potential innovations for their improvements. Source: Jens Walter’s presentation at the GMFH World Summit 2026.
Diet signatures in the gut microbiome are associated with gut and metabolic health outcomes
Personalized medicine and nutrition based on the human metabolome and gut microbiome were central topics at the conference. Eran Segal, PhD, a computer scientist at the Weizmann Institute (Israel), presented groundbreaking research from the Human Phenotype project showing that people respond to the same foods in vastly different ways. Segal’s team also used simulated models to demonstrate how personalized diets can predict shifts in the gut microbiome that lead to better cardiometabolic health11. Taken together, these findings illustrate that individual responses to food depend on gut microbiome composition and the metabolites gut microbes and human cells produce. So, nutrient needs and the benefits we obtain from diet are not one-size-fits-all.
Nicolata Segata, PhD, from the CIBIO Department at the University of Trento (Italy), presented data from the PREDICT UK and US cohorts showing species-level diversity of the microbiome partly explains the personal response to food12. On the other hand, findings from the PROSPECT study showed stool metagenomics had a strong screening capacity for colorectal cancer as compared to prediabetes and diabetes13. When it comes to microbiome acquisition and transmission, Segata acknowledged that the gut microbiome is largely transmitted horizontally, with food categories harboring distinct microbial traits that shape food-to-gut transmission and intestinal colonization14.

Sources of human microbiome acquisition and transmission. Source: Nicola Segata’s presentation at the GMFH World Summit 2026.
The gut microbiome mirrors dietary long-term patterns, with some gut microorganisms being a hallmark of a healthy microbiome. Francesco Asnicar, PhD, from the CIBIO Department at the University of Trento (Italy), acknowledged that the gut parasite Blastocytis is more prevalent in the gut microbiome than previously thought, with Fiji exhibiting the highest prevalence (56.3%) while Japan having the lowest (2.5%). Higher carriage of Blastocystis was linked to healthier plant-based diets and favorable cardiometabolic biomarkers15. Subtypes of Blastocystis varied by region, with subtypes 1 and 2 dominating in non-Westernized populations and subtype 4 more common in Europe. These findings position Blastocystis as a marker for gut and metabolic health, challenging its traditional classification as a pathogen.
Moving beyond fecal microbiota transfers towards targeted microbiome-based interventions
The central topic of a plenary session was what is new and relevant for clinical practice on gut microbiome-derived treatments. Max Nieuwdorp, PhD, a gastroenterologist at the Academic Medical Center in Amsterdam (The Netherlands), shared recent findings on a very strong correlation between post-prandial plasma ethanol and small intestinal lactic acid bacteria. Endogenous alcohol levels were about 50 times higher in portal vein blood of patients with metabolic dysfunction-associated steatotic liver disease (MASLD). However, antibiotics blocked liver alcohol dehydrogenase function and eradicated ethanol production in the human gut16. Nieuwdorp also acknowledged the potential benefits of fecal microbiota transfers (FMTs) on human obesity based on the available five clinical trials. Host, type of FMT (fresh or capsules), and presence of previously present strains in the gut appeared to be important determinants of the success of FMT engraftment in the recipient host17. The role of the gut microbial-derived metabolite 6-bromotryptophan is being explored on glucose levels in 36 people with metabolic syndrome from the BROMO trial, based on previous findings showing this microbiota-derived metabolite linked to preserved residual beta cell function18.
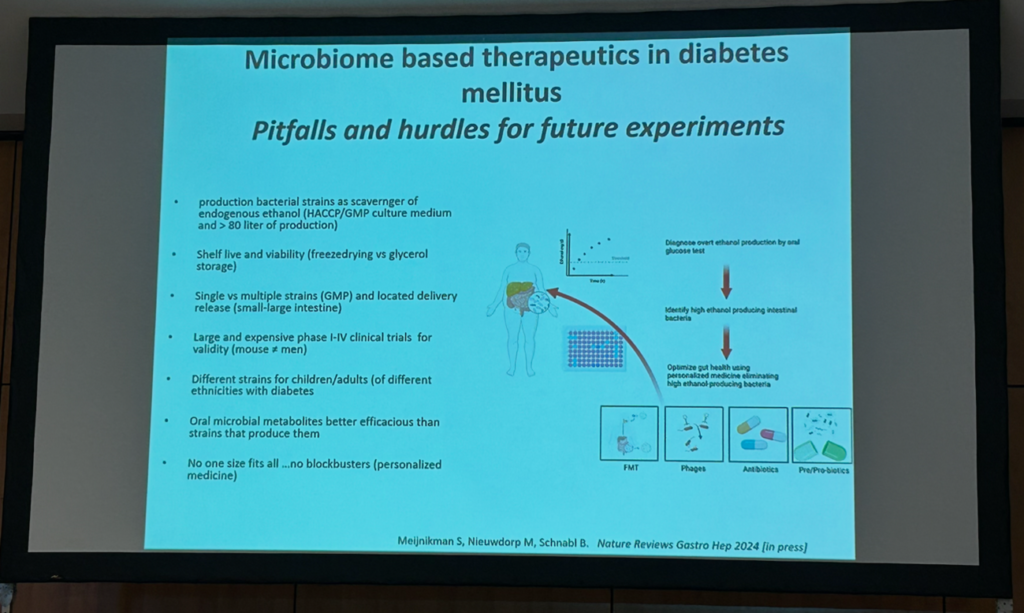
Pros and cons of microbiome therapeutics to target excessive ethanol production by gut bacteria. Source: Nieuwdorp’s presentation at the GMFH World Summit 2026.
Nicolas Benech, MD, PhD, from the Hôpital de la Croix-Rousse in Lyon (France), presented novel findings on the LIVEDIFF phase 1 trial demonstrating the safety of EXL01, a single isolated unmodified strain of Faecalibacterium prausnitzii, in preventing Clostridioides difficile infection (CDI) recurrence in patients at high risk. No treatment-related safe adverse effects were reported at 8 weeks (primary outcome). Interestingly, the success rate (no CDI recurrence within 8 weeks) was similar between EXL01 (83.3%) and FMT-treated patients matched on age, sex, and prior CDI-therapy (79.4%) from the French Faecal Transplant Group cohort19. Benech acknowledged an association between EXL01 in feces with butyrate and bile acids, showing that ecosystem-level functional recovery, rather than microbiome composition alone, is key in shaping the efficacy of live biotherapeutic products. The phase II trial is currently under recruitment and will include 50 patients in two arms (EXL01 and placebo).
Purna C. Kashyap, MBBS, AGAF, from the Mayo Clinic (US), updated how interconnected gut functions and the gut microbiome drive symptoms in IBS, including abnormal intestinal secretion, heightened visceral sensation, low-grade inflammation, increased intestinal permeability, and altered gut-brain communication. Kashyap presented unpublished findings in mice on particular gut microbial-derived metabolites that can shape how physiological stimuli would induce symptoms that could also be relevant for unexplained abdominal symptoms in IBS. Cooperative microbial signaling occurs through cellular integration hubs to modulate gut physiology, which in turn is modulated by diet and the overall metabolite makeup in the gut. For instance, the balance of host- and microbiome-derived metabolites hypoxanthine, butyrate, tryptamine, and primary bile acids was disrupted in IBS and may affect a distinct function (effectors) or broadly change responses to other metabolites (regulatory)20. Kashyap highlighted that IBS clinical trials and management need to evolve from symptom modulation to targeting complex pathophysiological mechanisms and the overall metabolite landscape21.
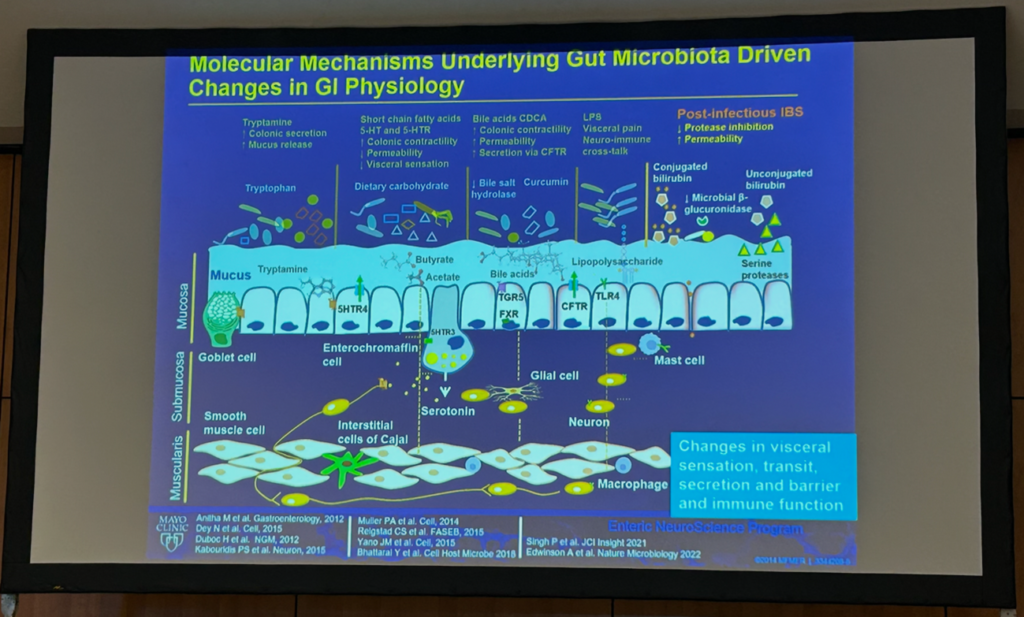
Integrating comprehensive clinical phenotyping with multi-omics allow to define how microbial metabolites influence host physiology. Source: Kashyap’s presentation at the GMFH World Summit 2026.
Simon Mark Dahl Baunwall, MD, PhD, from Aarhus University Hospital (Denmark), updated the role of FMT in IBD and IBS. Based on negative and inconclusive findings of FMT for ulcerative colitis, Crohn’s disease, and IBS, Dahl highlighted that the clinical effects of FMT might require donor strains with specific functional capacities (e.g., gas metabolism) that can successfully colonize the recipient. Other important factors that need to be considered are integrating microbial ecology to improve the recipient’s response to treatment, the potential use of FMT as a maintenance therapy, and how FMT can be integrated within treatment hierarchies established for GI disorders (e.g., FMT after advanced therapy for UC and FMT as a complementary approach to the low FODMAP diet).
Harnessing the gut microbiome for cancer therapy
The last plenary session covered recent landmark trials confirming that FMT is a promising approach to enhancing immunotherapy efficacy in advanced solid tumors. Gianluca Ianiro, MD, PhD, a gastroenterologist from the Fondazione A. Gemelli IRCCS and Università Cattolica del Sacro Cuore in Rome (Italy), presented new findings from the randomized, double-blind placebo-controlled phase 2a TACITO trial supporting the safety and achievement of 12-month progression-free survival and median progression-free survival of selected donor FMT in patients with metastatic renal cell carcinoma with immune checkpoint inhibitors (ICIs)22. Unpublished data showed that higher levels of donor-derived and endogenous Akkermansia in the gut were associated with improved survival outcomes. These findings are promising considering that the efficacy of monotherapy with ICIs rarely exceeds 40%. Ianiro also acknowledged some issues with FMT, including its lack of precision and scalability, especially if FMT should be repeated over time23. Ianiro also presented best practices on gut microbiome testing in clinical practice based on an international consensus to regulate gut microbiome testing24.
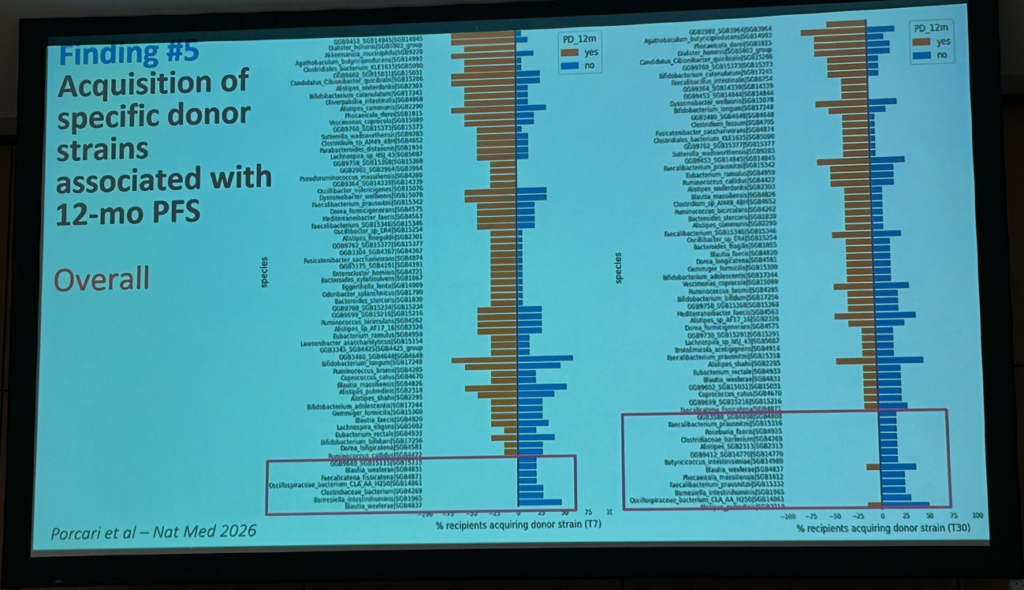
Acquisition of specific donor strains is associated with 12-month progression-free survival in treatment-naïve patients with metastatic renal cell carcinoma receiving pembrolizumab and axitinib. Source: Ianiro’s presentation at the GMFH World Summit 2026.
Lisa Derosa, MD, PhD, from Gustave Roussy (France), presented recent findings from the Oncobiome project focusing on antibiotic-related gut dysbiosis in oncology as a risk factor for resistance to ICI therapies. Akkermansia muciniphila emerged as a potential marker in predicting clinical response to immunotherapies in patients with advanced non-small-cell lung cancer (NSCLC), while pro-inflammatory Enterocloster and Clostridium taxa were the main ecological drivers of dysbiosis25,26. Derosa also announced the first IMMUNOLIFE phase 2 study to explore the potential of an oral microbiome therapy (MaaT033) in combination with cemiplimab versus best investigator choice (second-line treatment), in patients with advanced NSCLC who present antibiotic-induced gut dysbiosis.
Miguel Zugman, MD, from City of Hope Comprehensive Cancer Center (US), shared new findings from randomized phase 1 trials showing that supplementation with CBM588, a probiotic strain of Clostridium butyricum, may augment the response to checkpoint inhibitors and tyrosine kinase inhibitors in patients with kidney cancer27,28. Other ongoing microbiome research in these patients includes exploring the role of adding a prebiotic named Camu Camu to enhance the efficacy of immunotherapy. Another single bacterial strain with promising effects in boosting the efficacy of ICIs is EXL01, which restored the anti-tumor response to ICI in the context of antibiotic-induced microbiome disturbance at clinical and tumor transcriptomics level. While EXL01 strain did not induce any change in fecal microbiome, transcriptomic analysis showed it had a direct effect on immune cells in the small intestine29. Zugman acknowledged that EXL01 will be evaluated as an adjuvant to ICI in multiple cancers.
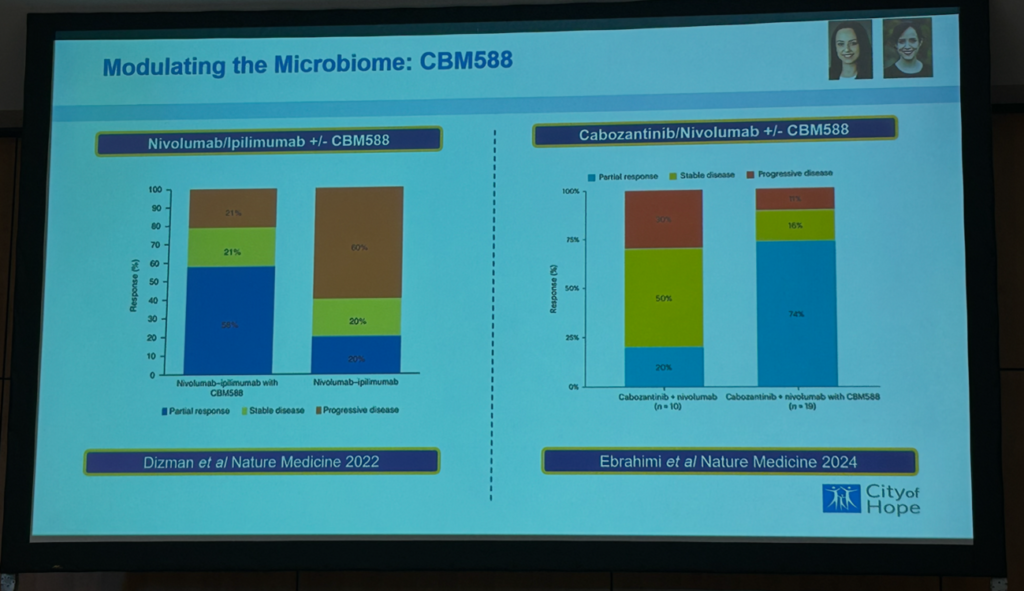
A probiotic supplement of Clostridium butyricum boosts response to cancer immunotherapy. Source: Zugman’s presentation at the GMFH World Summit 2026.
Diet and nutrition in IBD, liver diseases, IBS, and cancer: from bench to bedside
Beyond plenary sessions, four parallel workshop sessions took place during the GMFH World Summit. A first roundup of workshops covered the role of diet in IBD prevention and treatment based on the first consensus on diet and nutrition in IBD and the role of metabolic responses in the development of liver cancer and nutritional care as part of treatment of liver diseases. A second session of two parallel workshops focused on new advances in the pathogenesis of IBS and tips and tricks for dietary management of IBS based on etiology, as well as modulating diet and gut microbiome to enhance response and reduce toxicity to cancer treatment.
The 15th GMFH World Summit will take place in 2028 in Bordeaux (France).

Moro photos available at the ESNM website
Further reading:
- Petrone BL, Aqeel A, Jiang S, et al. Diversity of plant DNA in stool is linked to dietary quality, age, and household income. PNAS. 2023; 120(27):e2304441120. doi: 10.1073/pnas.2304441120.
- Whelan K, Bancil AS, Lindsay JO, et al. Ultra-processed foods and food additives in gut health and disease. Nat Rev Gastroenterol Hepatol. 2024; 21(6):406-427. doi: 10.1038/s41575-024-00893-5.
- Bancil A, Rossi M, Sandall A, et al. DOP097 Emulsifier restriction restriction is an effective therapy for active Crohn’s disease: the ADDapt trial – a multi-centre, randomised, double-blind, placebo-controlled, re-supplementation trial in 154 patients. J Crohns Colitis. 2025; 19(Suppl 1):i262. doi: 10.1093/eccojcc/jjae190.0136.
- Armstrong HK, Bording-Jorgensen M, Santer DM, et al. Unfermented b-fructan fibers fuel inflammation in select inflammatory bowel disease patients. Gastroenterology. 2023; 164(2):228-240. doi: 10.1053/j.gastro.2022.09.034.
- Tollenaar SL, Khorasaniha R, Jovel J, et al. Reduced fibre-fermenting capacity of gut microbes in multiple sclerosis may result in prebiotic dietary fibre b-fructan promoting inflammation and CNS damage. eGastroenterology. 2026; 4(1):e100296. doi: 10.1136/egastro-2025-100296.
- Oliero M, Hajjar R, Cuisiniere T, et al. Inulin impacts tumorigenesis promotion by colibactin-producing Escherichia coli in ApcMin/+ mice. Front Microbiol. 2023; 14:1067505. doi: 10.3389/fmicb.2023.1067505.
- Moerings BGJ, Abbring S, Tomassen MMM, et al. Rice-derived arabinoxylan fibers are particle size-dependent inducers of trained immunity in a human macrophage-intestinal epithelial cell co-culture model. Curr Res Food Sci. 2023; 8:100666. doi: 10.1016/j.crfs.2023.100666.
- Li F, Armet AM, Korpela K, et al. Cardiometabolic benefits of a non-industrialized-type diet are linked to gut microbiome modulation. Cell. 2025; 188(5):1226-1247.e18. doi: 10.1016/j.cell.2024.12.034.
- Armet AM, Deehan EC, Thöne JV, et al. The effect of isolated and synthetic dietary fibers on markers of metabolic diseases in human intervention studies: a systematic review. Adv Nutr. 2020; 11(2):420-438. doi: 10.1093/advances/nmz074.
- Armet AM, Li F, Deehan EC, et al. Mechanistic insights into microbiome-dependent and personalized responses to dietary fibre in a randomized controlled trial. medRxiv [Preprint]. 2025. doi: 10.1101/2025.11.20.25340625.
- Segev T, Barak D, Zahavi L, et al. Diet-microbiome associations in 10,068 individuals from the Human Phenotype Project to guide personalized nutrition. Nat Med. 2026. doi: 10.1038/s41591-026-04312-x.
- Valles-Colomer M, Menni C, Berry SE, et al. Cardiometabolic health, diet and the gut microbiome: a meta-omics perspective. Nat Med. 2023; 29(3):551-561. doi: 10.1038/s41591-023-02260-4.
- Piccinno G, Thompson KN, Manghi P, et al. Pooled analysis of 3,741 stool metagenomes from 18 cohorts for cross-stage and strain-level reproducible microbial biomarkers of colorectal cancer. Nat Med. 2025; 31(7):2416-2429. doi: 10.1038/s41591-025-03693-9.
- Carlino N, Blanco-Míguez A, Punčochář M, et al. Unexplored microbial diversity from 2,500 food metagenomes and links with the human microbiome. Cell. 2024; 187(20):5775-5795.e15. doi: 10.1016/j.cell/2024.07.039.
- Piperni E, Nguyen LH, Manghi P, et al. Intestinal Blastocystis is linked to healthier diets and more favorable cardiometabolic outcomes in 56,989 individuals from 32 countries. Cell. 2024; 187(17):4554-4570.e18. doi: 10.1016/j.cell.2024.06.018.
- Meijnikman AS, Davids M, Herrema H, et al. Microbiome-derived ethanol in nonalcoholic fatty liver disease. Nat Med. 2022; 28(10):2100-2106. doi: 10.1038/s41591-022-02016-6.
- Schmidt TSB, Li SS, Maistrenko OM, et al. Drivers and determinants of strain dynamics following fecal microbiota transplantation. Nat Med. 2022; 28(9):1902-1912. doi: 10.1038/s41591-022-01913-0.
- de Groot P, Nikolic T, Pellegrini S, et al. Faecal microbiota transplantation halts progression of human new-onset type 1 diabetes in a randomised controlled trial. Gut. 2021; 70(1):92-105. doi: 10.1136/gutjnl-2020-322630.
- Benech N, Cassir N, Alric L, et al. Impact of clinical and pharmacological parameters on faecal microbiota transplantation outcome in Clostridioides difficile infections: results of a 5-year French national survery. Aliment Pharmacol Ther. 2025; 61(1):159-167. doi: 10.1111/apt.18330.
- Liu S, Domingo-Vidal M, Madhu B, et al. Metabotherapy for intestinal disease: using metabolites to prevent and treat disorders of the gut. Nat Rev Gastroenterol Hepatol. 2026. doi: 10.1038/s41575-026-01178-9.
- Mars RAT, Frith M, Kashyap PC. Functional gastrointestinal disorders and the microbiome-what is the best strategy for moving microbiome-based therapies for functional gastrointestinal disorders into the clinic? Gastroenterology. 2021; 160(2):538-555. doi: 10.1053/j.gastro.2020.10.058.
- Porcari S, Ciccarese C, Heidrich V, et al. Fecal microbiota transplantation plus pembrolizumab and axitinib in metastatic renal call carcinoma: the randomized phase 2 TACITO trial. Nat Med. 2026. doi: 10.1038/s41591-025-04189-2.
- Danne C, Rolhion N, Sokol H. Recipient factors in faecal microbiota transplantation: one stool does not fit all. Nat Rev Gastroenterol Hepatol. 2021; 18(7):503-513. doi: 10.1038/s41575-021-00441-5.
- Porcari S, Mullish BH, Asnicar F, et al. International consensus statement on microbiome testing in clinical practice. Lancet Gastroenterol Hepatol. 2025; 10(2):154-167. doi: 10.1016/S2468-1253(24)00311-X.
- Derosa L, Routy B, Maltez Thomas A, et al. Intestinal Akkermansia muciniphila predicts clinical response to PD-1 blockade in patients with advanced non-small-cell lung cancer. Nat Med. 2022; 28(2):315-324. doi: 10.1038/s41591-021-01655-5.
- Allan Almonte A, Thomas S, Iebba V, et al. Gut dysbiosis in oncology: a risk factor for immunoresistance. Cell Res. 2026; 36(2):103-120. doi: 10.1038/s41422-025-01212-6.
- Dizman N, Meza L, Bergerot P, et al. Nivolumab plus ipilimumab with or without live bacterial supplementation in metastatic renal cell carcinoma: a randomized phase 1 trial. Nat Med. 2022; 28(4):704-712. doi: 10.1038/s41591-022-01694-6.
- Ebrahimi H, Dizman N, Meza L, et al. Cabozantinib and nivolumab with or without live bacterial supplementation in metastatic renal cell carcinoma: a randomized phase 1 trial. Nat Med. 2024; 30(9):2576-2585. doi: 10.1038/s41591-024-03086-4.
- Bredon M, Danne C, Phuong Pham H, et al. Faecalibacterium prausnitzii strain EXL01 boosts efficacy of immune checkpoint inhibitors. Oncoimmunology. 2024; 13(1):2374954. doi: 10.1080/2162402X.2024.2374954.

