Celiac disease affects approximately 1% of the population, although a much higher percentage of the population carry the genetic risk factors and consume gluten, the disease driving factor. The gut microbiota is emerging as an important cofactor, that could help explain why some genetically predisposed people develop celiac disease and others do not. The only current treatment for celiac disease is a strict gluten-free diet for life, which is difficult to follow, expensive, and doesn’t always lead to complete resolution of symptoms and inflammation. Better understanding of the relatively unknown role of small intestinal microbiota in celiac disease could help us prevent or better treat established disease.
Research groups have invested effort in identifying microbiota differences in cross-sectional studies in celiac patients versus healthy controls and in longitudinal studies where “at risk” individuals are followed up to until they develop celiac specific antibodies. Similar to what has been reported in other chronic inflammatory diseases, the microbial changes associated with celiac disease vary between studies and is not always consistent across studies.
According to Dr. Marco Constante from McMaster University (Hamilton, Canada) this is not surprising; “To date, the studies investigating the gut microbiota in celiac disease have varied in design, sampling location, and control populations.” In addition, most studies have looked at the microbiota composition in stool. “Inflammation in celiac disease primarily occurs in the upper small intestine known as the duodenum, and microbial changes that occur in feces may not reflect the changes that occur in the small intestine. This makes it difficult to gain insight into potential mechanisms at the site of disease” according to Dr. Constante.
A new study published in Gastroenterology shows gastrointestinal location is a key determinant of microbiota composition and function in celiac disease.
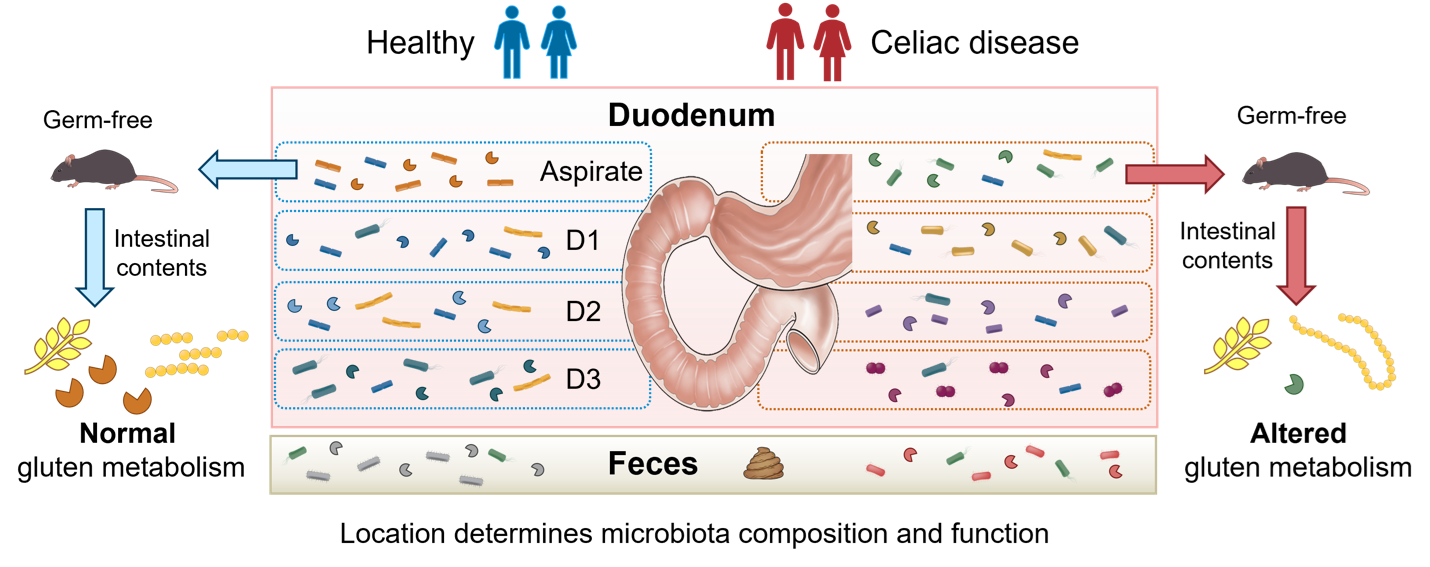
The study led by Dr. Alberto Caminero and Dr. Elena Verdu at McMaster University set out to analyze the gut microbiota along the gastrointestinal tract in patients with active celiac disease compared with healthy individuals. They studied the microbial composition and function in biopsies from different sections of the duodenum, as well as small intestinal aspirates and feces.
Location along the gastrointestinal tract, rather than the presence or absence of celiac disease, was the strongest determinant of microbiota composition. However, microbial differences were indeed detected between celiac patients and controls, that were specific to each location. Importantly, the microbiota composition in biopsies from the duodenum clustered according to the severity of inflammation. Neisseria, an opportunistic pathogen previously found to be increased in patients with active celiac disease, was associated with more severe enteropathy.
Even within different portions of the duodenum, location specific differences between patients with celiac disease and controls were observed, highlighting the importance of reporting precise sampling location in studies. Duodenum-specific differences were also observed when microbiota function was analyzed revealing altered microbial proteolytic function in patients with celiac disease.
It is known that proteases from bacteria can digest gluten, increasing or decreasing gluten immunogenicity. Indeed, microbial gluten-degrading enzymes such as novel computationally designed endopeptidases and latiglutenase are currently under investigation in clinical trials with promising results for improving gluten-induced symptoms and consequent quality of life.
The Constante et al. study also investigated mechanisms using germ-free mice that were colonized with small intestinal microbiota from celiac patients. These mice developed impaired capacity to digest gluten paralleled by higher detection of immunogenic gluten peptides in the intestine. Expression of microbial glutamate carboxypeptidase was lower in the duodenum of celiac patients, and its decrease in mice also correlated with impaired gluten degradation, suggesting a potential candidate for future therapeutic development.
“This is one of the first studies to perform a comprehensive comparison of the small intestinal and fecal microbiota in celiac disease, allowing us to identify specific microbial niches in celiac disease” says Dr. Caminero, an assistant professor at McMaster University and co-lead of the study. Stool is often used to investigate microbiota composition in celiac disease because its easily obtained from study participants; however, the changes seen in stool were not always observed in the small intestine. “The results from our study highlight that sampling location should be considered a confounding factor when investigating a microbiota role in celiac disease,” adds Dr. Verdu, a professor from McMaster University and corresponding author of the study.
References:
Verdu EF, Schuppan D. Co-factors, Microbes, and Immunogenetics in Celiac Disease to Guide Novel Approaches for Diagnosis and Treatment. Gastroenterology. 2021 Nov;161(5):1395-1411.e4. doi: 10.1053/j.gastro.2021.08.016. Epub 2021 Aug 17. PMID: 34416277.
Constante M, Libertucci J, Galipeau HJ, et al. (in press). Gastroenterology. 2022.
Caminero A, Galipeau HJ, McCarville JL, Johnston CW, Bernier SP, Russell AK, Jury J, Herran AR, Casqueiro J, Tye-Din JA, Surette MG, Magarvey NA, Schuppan D, Verdu EF. Duodenal Bacteria From Patients With Celiac Disease and Healthy Subjects Distinctly Affect Gluten Breakdown and Immunogenicity. Gastroenterology. 2016 Oct;151(4):670-83. doi: 10.1053/j.gastro.2016.06.041. Epub 2016 Jun 30. PMID: 27373514.
Pultz IS, Hill M, Vitanza JM, Wolf C, Saaby L, Liu T, Winkle P, Leffler DA. Gluten Degradation, Pharmacokinetics, Safety, and Tolerability of TAK-062, an Engineered Enzyme to Treat Celiac Disease. Gastroenterology. 2021 Jul;161(1):81-93.e3. doi: 10.1053/j.gastro.2021.03.019. Epub 2021 Mar 17. PMID: 33741317.
Syage JA, Green PHR, Khosla C, Adelman DC, Sealey-Voyksner JA, Murray JA. Latiglutenase Treatment for Celiac Disease: Symptom and Quality of Life Improvement for Seropositive Patients on a Gluten-Free Diet. GastroHep. 2019 Nov;1(6):293-301. doi: 10.1002/ygh2.371. Epub 2019 Oct 8. PMID: 32313451; PMCID: PMC7169937.