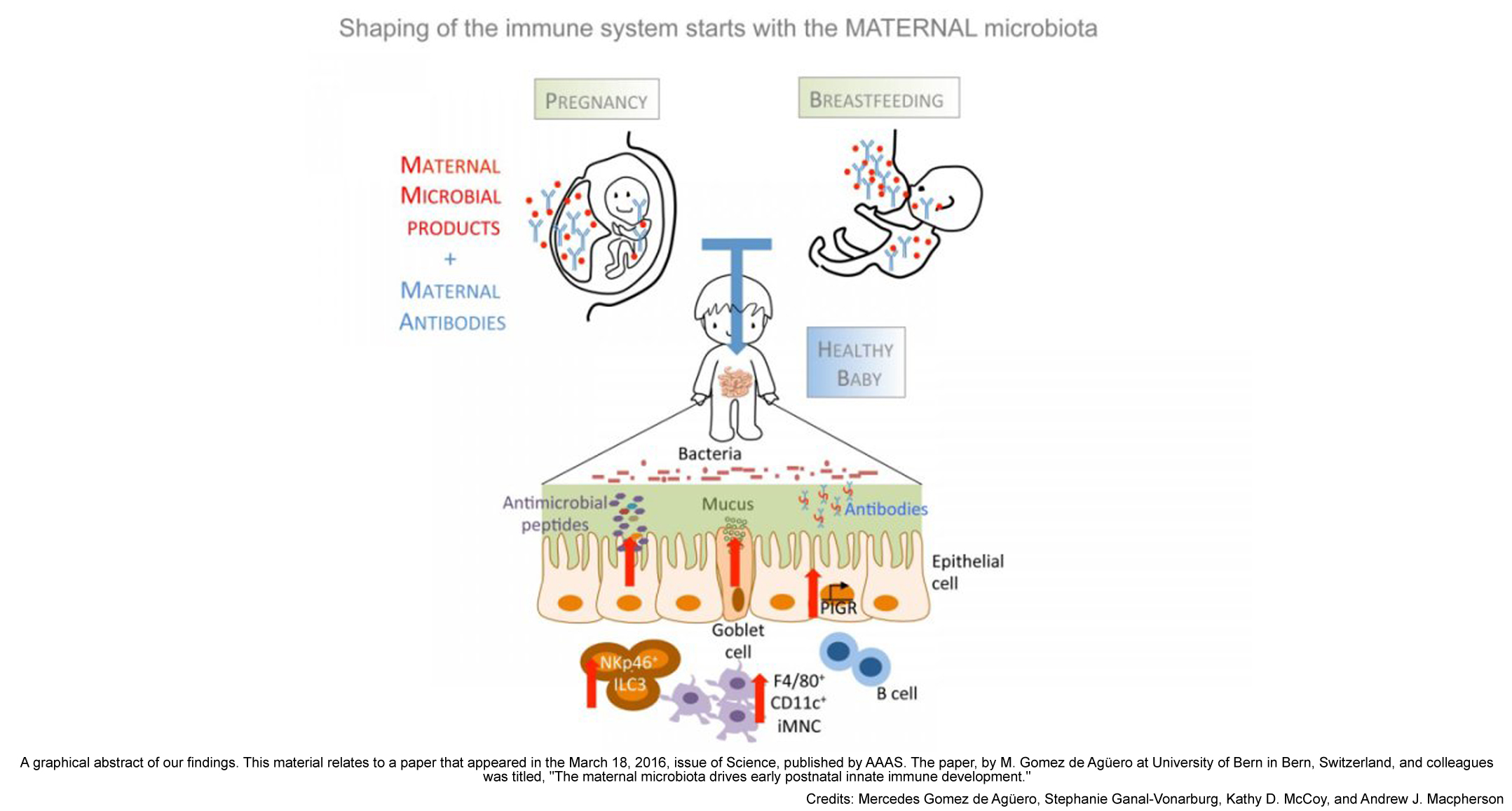
A recent study, led by Prof. Andrew Macpherson from the University of Bern (Switzerland), has found in mice that maternal microbiota and immunity strengthen the immune system of the newborn.
Although it is a well-known fact that a newborn’s gut microbiota can affect its own immune system, little is known about the impact of the maternal microbiota on her offspring’s immune development. By transiently colonizing pregnant female mice with gut bacteria, Gomez de Agüero et al. studied whether the maternal microbiota in pregnancy affects the early postnatal immune system of the offspring. The researchers used a system in which pregnant females were transiently colonized with genetically engineered Escherichia coli HA107, a strain that dwindles over time in the intestine and thus allows pregnant dams to become germ-free again before they give birth.
Compared to offspring born to germ-free female mice, the pups born to colonized mothers showed increased numbers of certain innate leukocytes and different patterns of gene expression in their guts. Maternal microbial exposure during pregnancy increased small intestinal innate lymphoid cell (ILC) and intestinal mononuclear cell (iMNC) proportions and total numbers compared with germ-free controls. This increase reached a maximum at 14 and 21 days after birth and persisted until at least 8 weeks of age in the colon.
Similar effects on early postnatal innate leukocytes were seen when pregnant dams were temporarily colonized with a cocktail of eight other microbes. After gestation-only colonization, B and T cell relative or absolute populations were not affected. Interestingly, maternal gestational colonization caused increased expression of several genes, including those that influence epithelial cell division and differentiation, mucus and ion channels, epithelial inmunoglobulin transport, and metabolism of dietary xenobiotics and bile acids, complex lipids, and sugars. These results suggest that the changes in mucosal transcriptional and metabolic signatures after maternal gestational colonization were part of a wide range of adaptations triggered by maternal exposure to intestinal microbes.
By transferring serum from bacteria-colonized pregnant mice to germ-free pregnant mice, the researchers found that maternal antibodies were required for gestational colonization effects. Maternal microbiota-derived compounds were transferred to maternal tissues and subsequently to the offspring and this process was increased in the presence of maternal antibodies. In addition, the researchers demonstrated that maternal antibodies enhanced the retention and transmission of microbial molecules. Maternal microbial molecules were bound to maternal IgG after intestinal exposure and therefore transferred to offspring across the placenta and through intestinal uptake from the milk. Maternal microbial molecules reached neonatal tissues such as the small intestine, liver, and spleen, and functionally protected them by limiting inflammatory responses to microbial molecules and penetration of intestinal microbes.
To sum up, during gestation, the maternal microbiota shaped the offspring’s immune system and prepared it for participating in a host-microbial mutual relationship. Maternal antibody-mediated transfer of bacterial products was required for gestational colonization effects.
Reference:
Gomez de Agüero M, Ganal-Vonarburg SC, Fuhrer T, et al. The maternal microbiota drives early postnatal innate immune development. Science. 2016;351(6279):1296-302.