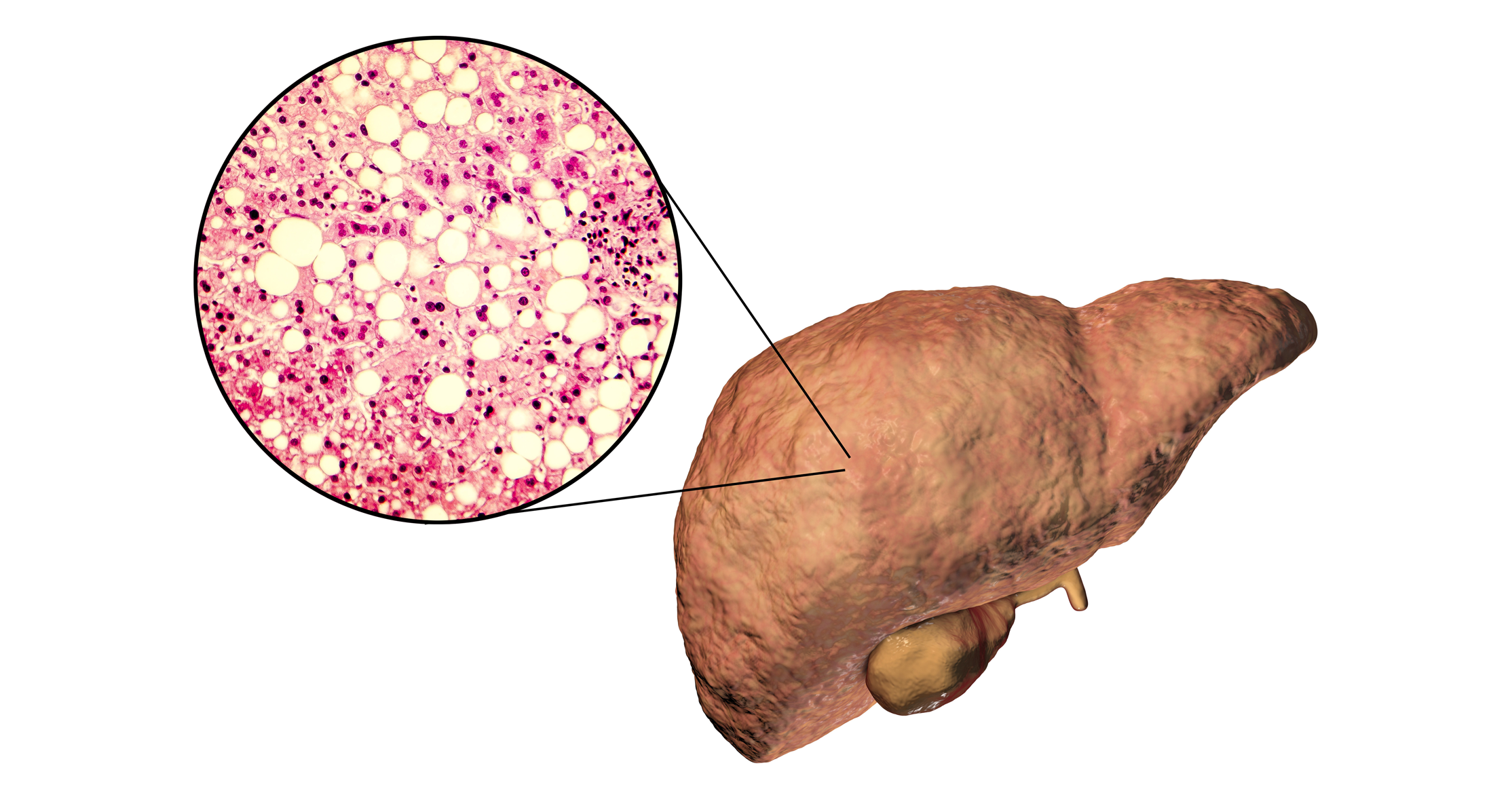
Non-alcoholic fatty liver disease (NAFLD) is the main cause of chronic liver disease in developed countries. NAFLD often shares symptoms with other metabolic disorders such as type 2 diabetes and obesity.
Recently, our gut microbiota has been revealed as a contributor to fatty liver disease development in both rodent models and NAFLD patients. However, the question of whether gut microbes make a causal contribution to the disease remains unknown.
Investigations led by Dr. Di Liu from the Wuhan Institute of Virology and University of Chinese Academy of Sciences in Beijing (China), indicated that the commensal alcohol-producing Klebsiella pneumoniae may accelerate the development of NAFLD, thus emerging as a new risk factor for this metabolic disease.
This research is based on an initial finding in an individual who, although not consuming alcohol, developed severe non-alcoholic steatohepatitis (NASH). A K. pneumoniae strain with the ability to produce alcohol endogenously was consequently identified and isolated as the contributor. Meanwhile, gut fungi were ruled out as contributors as anti-fungal treatments did not affect blood alcohol levels.
As a result of this case, it is possible that increased endogenous production of alcohol might be one mechanism by which the gut microbiota may drive the severity of NAFLD. In this regard, previous findings found that the serum alcohol concentration—mainly from an increased abundance of alcohol-producing Escherichia—was higher in NASH patients when compared with obese or control groups.
Yuan and colleagues were interested in validating the association between commensal alcohol-producing K. pneumoniae and NAFLD in a small cohort of Chinese patients with this disorder.
Sixty-one percent of patients with NAFLD carried high- and medium-alcohol-producing K. pneumoniae, compared with healthy controls, who harbored 6% of alcohol-producing K. pneumoniae. Furthermore, patients who recovered from NAFLD and lost weight after 6-9 months showed decreased fecal levels of alcohol-producing K. pneumoniae.
The authors then went a step further and explored the causal contribution of alcohol-producing K. pneumoniae in NAFLD.
Orally transferring patients’ isolates of high-alcohol-producing K. pneumoniae into mice induced NAFLD, which was aggravated when mice were fed a high-fat diet. An increase in the proportion of T helper 17 cells, neutrophils, macrophages and B cells—leading to a pro-inflammatory milieu—alongside mitochondrial dysfunction in the liver tissues of mice with NAFLD explained the contribution of alcohol-producing gut bacteria to the progression of fatty liver disease.
To the same extent, transplanting fecal microbiota from an individual with NASH into mice also induced NAFLD. In contrast, when the K. pneumoniae strain was selectively removed before fecal microbiota transplantation, the recipient mice did not develop NAFLD.
Finally, a set of mice experiments showed that alcohol was independently produced by the gut microbiota. Moreover, after the oral glucose tolerance test, NASH patients who showed a clinical improvement had lower blood alcohol concentration. Thus, it seems that blood alcohol concentrations after glucose intake could be a potential clinically non-invasive marker that reflects the gut levels of alcohol-producing K. pneumoniae and might help in predicting clinical improvement in patients with NAFLD.
Taken together, these findings suggest that endogenous alcohol produced by certain gut commensals might be another risk factor for NAFLD alongside current known risk factors for the disease. As such, this research will help identify new therapeutic targets that could improve quality of life and wellbeing for patients with the disease.
Reference:
Yuan J, Chen C, Cui J, et al. Fatty liver disease caused by high-alcohol-producing Klebsiella pneumoniae. Cell Metab. 2019; 30(4):675-88. doi: 10.1016/j.cmet.2019.08.018.